Linear
A linear shape is formed when there is no central atom OR when there are 2 electron pairs on the central atom and no lone pairs.


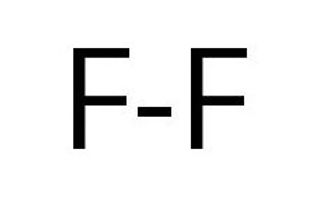



Take a look at these molecules (F2 and O2). They both are 2 atoms bonded together, therefore they have no central atom. Molecules with no central atom are always linear by default.We draw these molecules by using lines to show the bonds. A double line is used to show a double bond.
![]()
![]()

This is the BeH2 molecule. The central atom is beryllium with no lone pairs, and 2 electron pairs on it. This 3 atom molecule is also linear.
Trigonal Planar
A trigonal planar is formed when there are 3 electron pairs on the central atom and it has no lone pairs.



BF3 has 3 electron pairs around the central atom and no lone pairs. Therefore the atoms would spread out equally around the central atom, creating a trigonal planar.
Tetrahedral
A tetrahedral is formed when there are 4 electron pairs on the central atom and it has no lone pairs.



This molecule (CH4) has 4 electron pairs on carbon and no lone pairs. The hydrogens would spread out equally around carbon, creating a tetrahedral which is a 3D shape. The striped line shows that the atom is going behind, and a triangle is used to show that it is coming forward.
Trigonal Pyramidal
A trigonal pyramidal is formed when there are 4 pairs of electron pairs and 1 of those pairs is a lone pair.


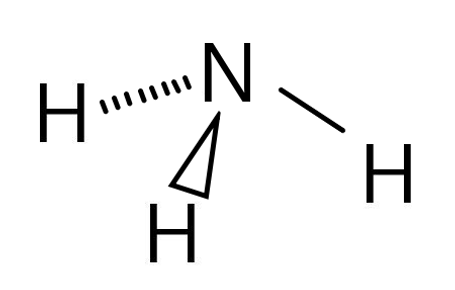
Ammonia (NH3) is trigonal pyramidal because there are 4 electron pairs, however one the those pairs is a lone pair (non-bonding). This molecule will look similar to a tetrahedral shape, but the lone pair will be invisible.
Linear Bent
A linear bent shape is formed when there are 4 pairs of electron pairs and 2 of those pairs are non-bonding.



Water, for example, is when 2 hydrogens are bonded with an oxygen, forming H20. Oxygen has 4 electron pairs, but 2 of the 4 pairs will be lone pairs. Again, this will result in a shape like the tetrahedral, but the 2 lone pairs will be invisible. This result in a 2D linear bent shape.